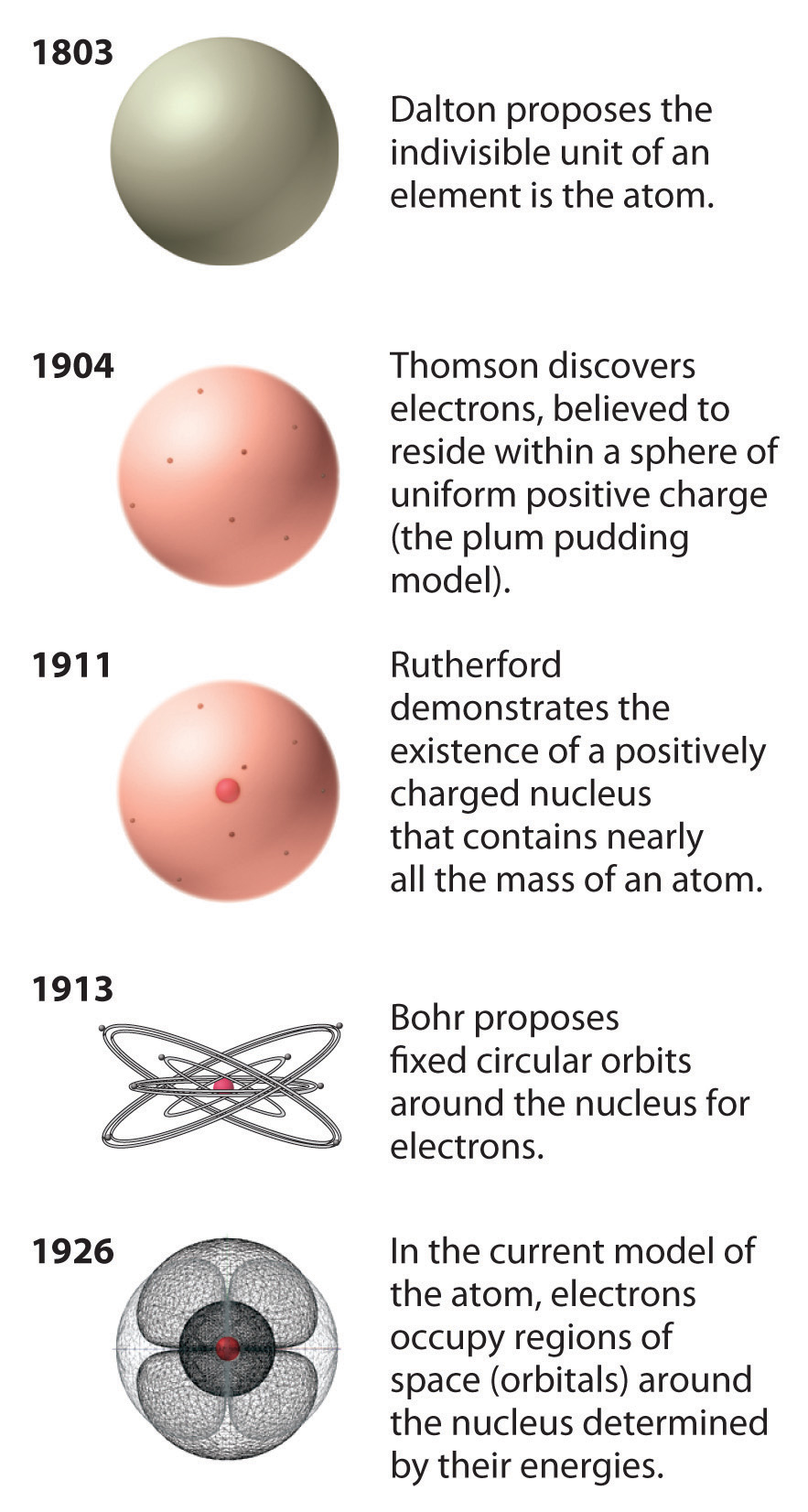
Bohr's model of atom3/6/2024 The emission spectrum of atomic hydrogen is divided into a number of spectral series, with wavelengths given by the Rydberg formula: \frac. This means that radiations possess both wave-like and particle-like. One of the concepts that played a major role in the formulation of Bohr’s model is the dual nature of electromagnetic radiation. Through his experiments, the scientist Neils Bohr improved upon Rutherford’s model of the atom. Hydrogen Spectrum: Spectral lines of Hydrogen, divided into series. Development Leading to Bohr’s Model of Atom. Similarly, the emission spectra of molecules can be used in chemical analysis of substances. Bohr’s model of the hydrogen atom, proposed by Niels Bohr in 1913, was the first quantum model that correctly explained the hydrogen emission spectrum. Each element’s emission spectrum is unique, and therefore spectroscopy can be used to identify elements present in matter of unknown composition. The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted by an atom’s electrons when they are returned to a lower energy state. Up to 2 electrons orbit in the first energy level. The different orbit distances are called energy levels. In the Bohr model of the atom: Electrons orbit the nucleus at different distances. He used the nuclear model to create his model.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
